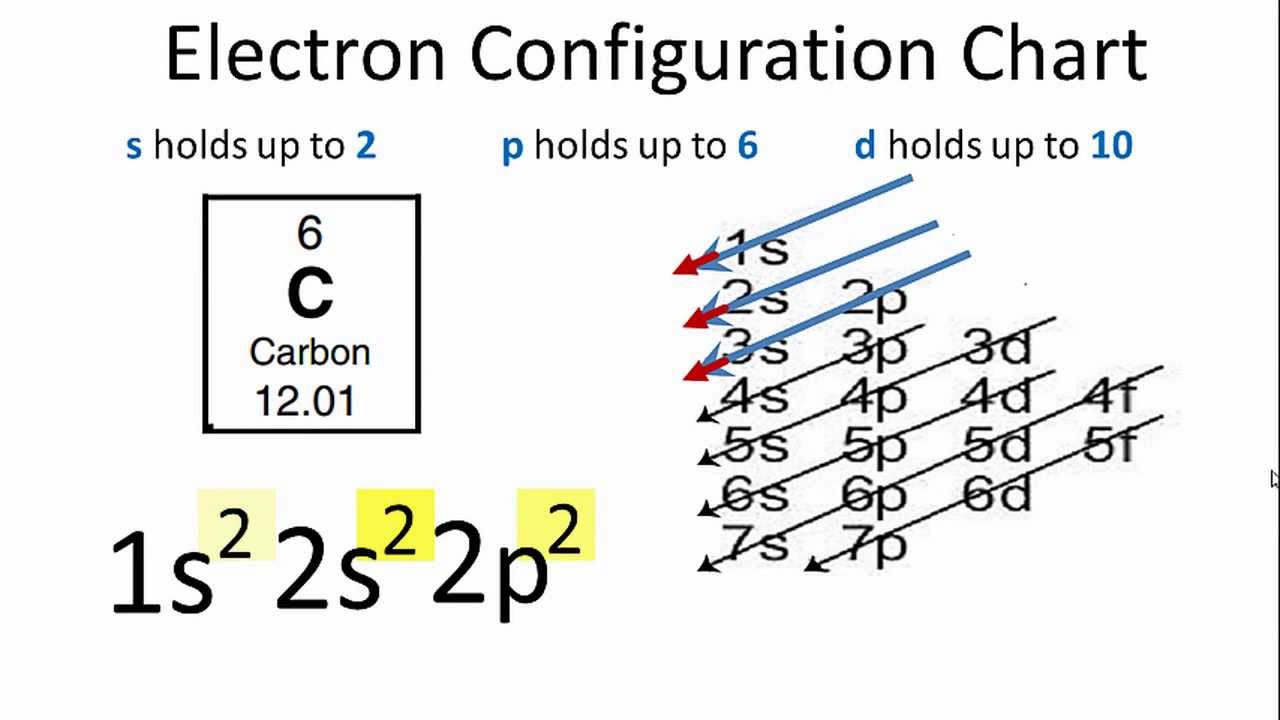 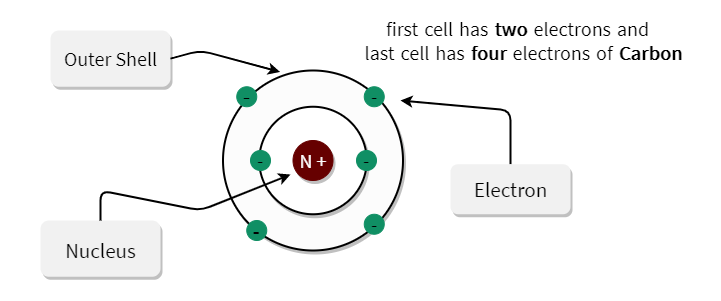
We can say that the valence electron of carbon is 4. you can add then you can see that there are 4 valence electron for the carbon atom in second energy level. Write the order of higest energy, for electron configuration of carbon atom. Therefore, we can write the electron configuration for carbon will be,įinding the number of valence electron for the carbon, only you look highest energy in electron configuration. but in case of carbon atom only need 2 electrons. and remaining 2 electrons for carbon atom will go in the 2p orbital. You know that, 2nd orbital has capable to hold only 2 electrons. and next 2 electrons for carbon atom will goes in the 2nd orbitals. since, 1s orbitals can only hold 2 electrons. we can write electron configuration for carbon atom, first 2 electrons will go in the first orbital. Now, if you loot at the electron configuration of carbon atom. Now, i m going to draw electron configuration for carbon atom. this is the number of porton.if neutral then we have 6 electron. if you look at the periodic table, you can see that the atomic number of carbon is 6. First need to draw the electron configuration of carbon atom. Here, i m going to discuss about how to identify valence electron for carbon atom. 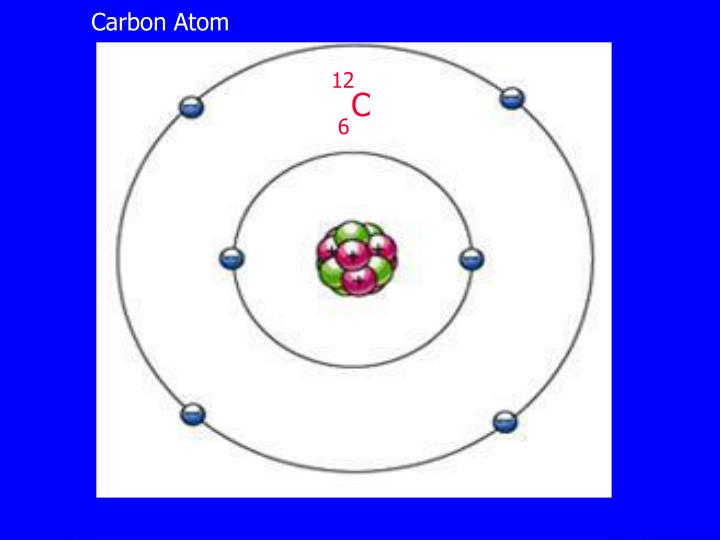
this is also easier techniques for calculating valence electron for an atoms. The other method is use to finding valence electron for any different atoms is, use the electron configuration. Arrange the atoms to show specific connections. Each hydrogen atom (group 1) has one valence electron, carbon (group 14) has 4 valence electrons, and oxygen (group 16) has 6 valence electrons, for a total of (2)(1) + 4 + 6 12 valence electrons. it mean, valence electron in outermost shell is 4. Determine the total number of valence electrons in the molecule or ion. The inner shell contain 2 electron out of 6. Lets know, with electron digram for carbon atom.Ītomic number of carbon atom is 6. 
this is the easiest and better method to find out the valence electron for any different atoms. so, according to above information, surely, we can say that carbon atom have 4 valence electrons. The first easiest methods for finding the number of valence electrons for carbon atom or any different atoms, first look at the periodic table, if you properly look at the periodic table then you can see that, carbon in group 14. (a) The positively charged carbon atom is surrounded by six valence electrons carbon has three valence electrons, and each hydrogen brings three valence. how many valence electrons does carbon have? First Method: Read More – What is the valency of sulphur? sulfur electron configuration. Therefore, place carbons in the center and hydrogen and oxygen on either side.How many valence electrons does carbon have – there are 4 valence electron in carbon atom. its valence electron with other carbon atoms or with atoms of other elements. Download scientific diagram 4: The behaviour of the valence electrons in carbon as a carbon atom is in free space and as multiple carbon atoms are brought. Let’s assume that the central atom is right carbon. Bonding in Carbon : A carbon atom has a total of six electrons carbon atom. Here, there are two carbon atoms, so we can assume any one as the central atom. Since carbon is less electronegative than oxygen, assume that the central atom is carbon. Place the least electronegative atom at the center. Now we have to choose the central atom from carbon and oxygen. Because the central atom is bonded with at least two other atoms, and hydrogen has only one electron in its last shell, so it can not make more than one bond. Here hydrogen can not be the central atom. So the total electron pairs = 24 ÷ 2 = 12 Total electron pairs = total valence electrons ÷ 2 And when we divide this value by two, we get the value of total electron pairs. Learn how to find: Carbon valence electrons, Hydrogen valence electrons, and Oxygen valence electrons So the total valence electrons = 8 + 3 + 12 +1 = 24 Now the CH 3COO – has a negative (-1) charge, so we have to add one more electron. Valence electrons of two carbon atoms = 4 × 2 = 8 Valence electrons of three hydrogen atoms = 1 × 3 = 3 Valence electrons of two oxygen atoms = 6 × 2 = 12 Since CH 3COO – has two carbon atoms, three hydrogen atoms, and two oxygen atoms, so… Hence, carbon has four valence electrons, hydrogen has one valence electron, and oxygen has six valence electrons. In the periodic table, carbon lies in group 14, hydrogen lies in group 1, and oxygen lies in group 16.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
